Leaky Gut vs. The Postbiotic Fortress: Strengthening Your Microbiome Shield |
|
|
|
Darrell Miller | 02/20/26 |
Date:
February 20, 2026 01:50 PM
Author: Darrell Miller
(support@vitanetonline.com)
Subject: Leaky Gut vs. The Postbiotic Fortress: Strengthening Your Microbiome Shield
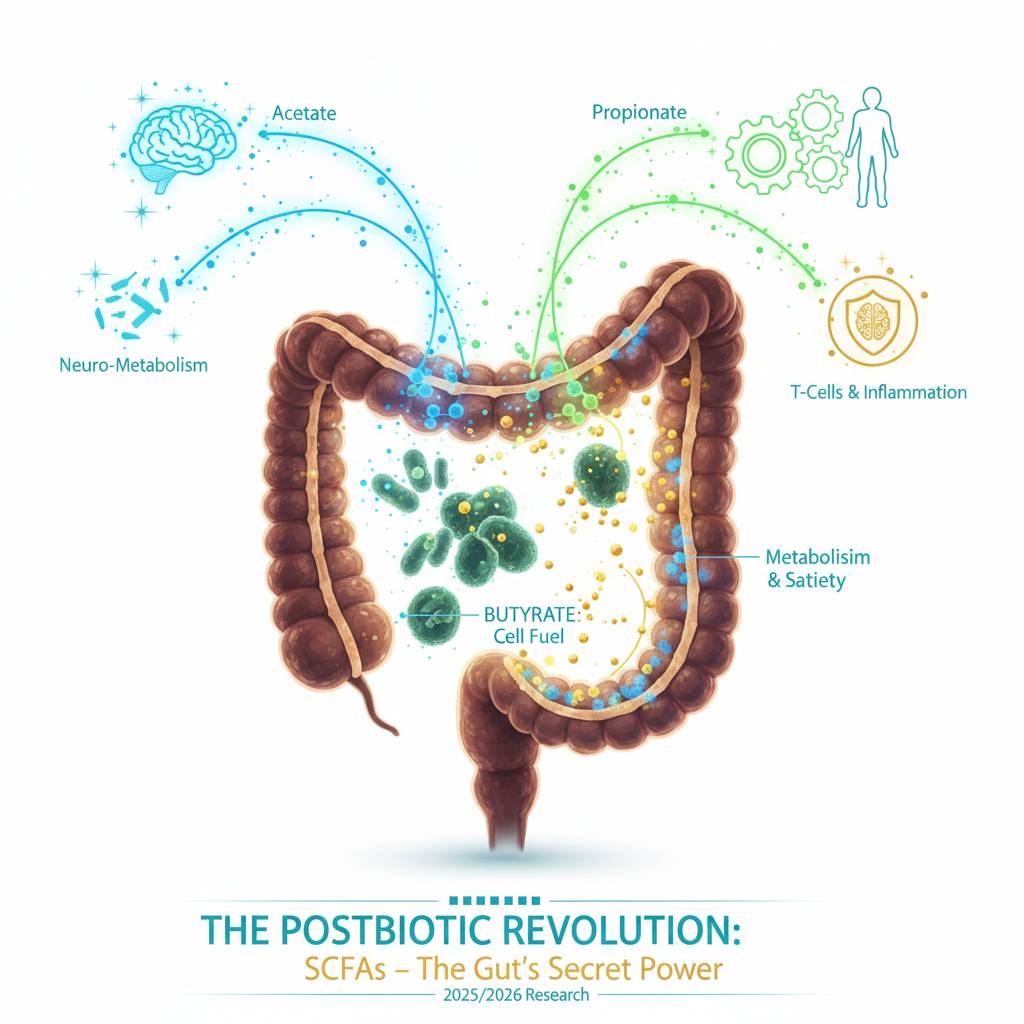
The "Postbiotic" Revolution: Why Your Gut’s Waste is Its Best Asset
For years, the conversation around gut health was a two-player game. You had Prebiotics (the fiber-rich "fuel") and Probiotics (the "live bacteria"). But in 2025 and 2026, the scientific community has pivoted to the third and most important piece of the puzzle: Postbiotics.While we’ve long obsessed over the "tenants" living in our gut, we are finally realizing that it’s their work - specifically the chemical metabolites they produce - that actually runs the show. Postbiotics, particularly Short-Chain Fatty Acids (SCFAs) like Butyrate, Propionate, and Acetate, are the actual "workers" that translate bacterial activity into human health.
The Big Three: Acetate, Propionate, and Butyrate
When you eat a high-fiber meal, your microbiome ferments those fibers into SCFAs. These aren't just waste products; they are high-powered signaling molecules that act like a Wi-Fi signal between your gut and the rest of your body.| Metabolite | Primary Role | System Targeted |
| Butyrate | Fuel & Barrier Integrity | Colon Health & Epigenetics |
| Propionate | Satiety & Liver Health | Metabolism & Appetite |
| Acetate | Systemic Energy & pH Balance | Brain & Weight Management |
Feeding the Gut from the Inside Out
Most cells in your body get their energy from the bloodstream. Your colonocytes (the cells lining your large intestine) are different. They are the only cells in the human body that prefer to eat "locally."Butyrate serves as the primary fuel source for these cells, providing up to 70% of their total energy requirements. Research from late 2025 emphasizes that without adequate butyrate, colon cells undergo a form of "cellular starvation," leading to a breakdown in the gut barrier—often referred to as "leaky gut." By "feeding" these cells from the inside, butyrate ensures the intestinal wall remains a tight, fortress-like seal against toxins.
2025/2026 Research: The Signaling Revolution
The most exciting shift in recent research (2025-2026) is the discovery of how these metabolites act as "systemic messengers." They don't just stay in the gut; they enter the bloodstream and talk to your most vital systems:- The Brain (Neuro-Metabolism): Studies published in mid-2025 have mapped the "SCFAs-microglia" pathway. It turns out SCFAs like Acetate can cross the blood-brain barrier to regulate neuroinflammation. This is opening new doors for treating "brain fog" and mood disorders by targeting the postbiotic profile.
- The Immune System: New 2026 clinical trials on postbiotic blends (such as the Postbiotic Active Lifestyle Blend) are showing that these metabolites can "prime" the immune system without the risks associated with live probiotics. They act as HDAC inhibitors, effectively "turning off" inflammatory genes in T-cells.
- Metabolism: Research from early 2026 indicates that Propionate is a heavy hitter for weight management. It signals the release of gut hormones like GLP-1 (the natural version of popular weight-loss drugs), naturally suppressing appetite and improving insulin sensitivity.
Why Postbiotics are the Future
The beauty of postbiotics lies in their stability. Unlike probiotics, which can die on the shelf or in your stomach acid, postbiotics are heat-stable and have a predictable impact.We are moving away from the "spray and pray" method of taking random bacteria and toward a precision model: delivering the specific metabolites your body needs to thrive.
The "Postbiotic" Revolution marks a pivotal shift in gut health, moving beyond live bacteria to focus on the powerful metabolites they produce, specifically Short-Chain Fatty Acids (SCFAs) like Acetate, Propionate, and Butyrate. While 2025/2026 research highlights how these molecules act as systemic messengers - regulating neuroinflammation in the brain and triggering natural satiety hormones like GLP-1 - the most critical scientific breakthrough remains the role of Butyrate as the primary energy source for colon cells. By fueling colonocytes from the inside out, these postbiotics maintain the integrity of the gut barrier and serve as the "actual workers" that translate microbial activity into tangible improvements for the immune system and metabolism.



